In the framework of the H2020 MSCA-ETN SULTAN project (https://etn-sultan.eu/), researchers from VITO and KU Leuven collaborated on a project aimed at developing an innovative solvometallurgical leaching method for extracting lead from zinc calcine and zinc leaching residue materials.
Potential and Challenge of Lead Extraction from Zinc Calcine and Zinc Leaching Residue
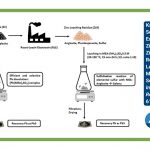
The extraction of lead from zinc calcine and zinc leaching residue poses both potential and challenges. The Roast-Leach-Electrowin (RLE) process is the predominant metallurgical method for generating over 95% of the current global zinc supply. This process starts with the roasting of zinc sulfide concentrate to yield a more reactive zinc oxide, known as zinc calcine.
Subsequently, zinc calcine, serving as an intermediary in the zinc industry, undergoes leaching in sulfuric acid to generate a zinc sulfate solution, which is further processed through electrowinning to yield pure zinc metal. Throughout this process, a significant amount of valuable metals, including lead, becomes entrapped in the residue, referred to as zinc leaching residue. Lead becomes immobilized within the residue as galena, exhibiting limited solubility in water-based systems.
Monoethanolamine Solvent Systems for Lead Extraction from Zinc Calcine and Zinc Leaching Residue
In this work the researchers of VITO and SIM2 KU Leuven developed an alternative solvometallurgical leaching based on monoethanolamine (MEA; H2NCH2CH2OH) to extract lead from zinc calcine and zinc leaching residue. MEA is a bifunctional organic solvent consisting of an amine and a hydroxyl functional group. For zinc calcine, a MEA-based lixiviant was applied to extract lead, thereby avoiding lead being locked and/or lost in zinc leaching residue after sulfuric acid leaching. Furthermore, in the second part of the study, the applicability of the MEA-based lixiviant was extended to the extraction of lead from zinc leaching residue material.
Utilizing the non-aqueous MEA-(NH4)2SO4 0.5 M system resulted in maximum lead leaching yield from zinc calcine (82%) after 12 hours of leaching at a mild temperature of 35 °C. FTIR analysis revealed the complexation process involving the interaction of metal ions with MEA molecules, while sulfate anions balanced the electrical charge of the cationic complex and stabilized the formed metal complexes.
Lead recovery was efficiently achieved through precipitation with Na2S, yielding galena (PbS) as the predominant precipitate. On the other hand, anglesite phases in the zinc leaching residue hindered lead extraction due to an instantaneous sulfidization reaction of the dissolved lead in the MEA-(NH4)2SO4 0.5 M system. Under basic MEA conditions, elemental sulfur naturally occurring in the zinc leaching residue formed sulfidizing species, facilitating the conversion of anglesite into insoluble galena.
Stability tests conducted by differential electrochemical mass spectroscopy (DEMS) and direct analysis in real-time mass spectroscopy (DART-MS) indicated the overall robust stability of the organic lixiviant, with no degradation products of MEA detected in either gas or liquid phases. In summary, assuming the absence of elemental sulfur in the initial material, the MEA-(NH4)2SO4 leaching system allows for efficient and selective lead extraction from anglesite (PbSO4), overcoming challenges associated with its limited solubility in water-based systems.
Full reference to the paper
Nor Kamariah, Panagiotis Xanthopoulos, Jia Song, Natalia Pires Martins, Jan Jordens, Koen Binnemans, Jeroen Spooren. Selective Lead Extraction from Zinc Calcine and Zinc Leaching Residue by Leaching with Monoethanolamine Solvent Systems. Ind. Eng. Chem. Res. 2024, 63, 14, 6110–6120. DOI: 10.1021/acs.iecr.4c00291.
Acknowledgements
The authors thank Myrjam Mertens and Kosar Hassannezhad for their help with XRD and Rietveld analysis. The authors also thank Dirk Vanhoyweghen for the help with polished sections, and Nancy Dewit for the help with SEM analysis. The research leading to these results has received funding from the European Community’s Horizon 2020 Program under Grant Agreement no. 812580 (MSCA-ETN SULTAN). This publication reflects only the authors’ view, exempting the Community from any liability.